by
Michael A. Pinto, CSP, CMP
A growing consideration when evaluating the quality of indoor environments is the role that building finishes, furnishings, and cleaning practices have on the transmission of infectious agents. This is especially true in medical facilities where health care acquired infections (HCAI) are having a growing impact on patient survival rates and health care costs. Anywhere from 48,000 to 100,000 fatalities annually can be tracked back to the development of infections in patients that did not have an infection upon admission.(1) Beyond the loss of life, these infections are expensive. Various researchers estimate that the extra days spent in the hospital lead to $8.1 billion in added costs.(2)
The move to address such problems has intensified since Medicare and Medicaid implemented a policy of refusing to reimburse health care facilities for certain preventable infections. Private insurance is now following their lead. This push toward recognizing infectious contaminants as part of the indoor environment has also grown as the European concept of “baubiology” has caught hold in North America.(3)
The move to new types of cleaning chemicals, including botanicals and “natural” cleaners from plant oils is one response to this deadly and expensive problem. But this change toward stronger and stronger chemicals for cleaning purposes generally only provides short-term relief as the side effects of harsh chemicals and the natural ability of bacteria and other infectious agents to develop a resistance to antimicrobial agents limits their effectiveness, especially in health care facilities.
Ultrasonic Cleaning to Control Infections?

Fortunately, there is an option that has been proven to dramatically enhance normal cleaning procedures in health care facilities: Ultrasonics. Ultrasonic cleaning kills bacteria and other pathogens through physical means rather than by a chemical reaction, eliminating the need for harsh compounds and the possibility of instigating resistant organisms.
After more than two years of testing in both the United States and Great Britain, the results showed conclusively that Morantz ultrasonic cleaning equipment was capable of removing bacterial contamination from items typically used in both institutional and residential settings. Over the course of three carefully controlled studies it was discovered that the Morantz ultrasonic machines were effective in:
- Removing both gross contamination and microscopic bacterial pathogens that are found on items after they have been involved in a black water loss (sewage).
- Destroying bacterial that pose the greatest concern to health professionals, with a strain of methicillin-resistant Staphylococcus aureus (MRSA) chosen to serve as a surrogate for such serious strains of microbial contamination.
- Supplementing normal cleaning procedures in hospitals to significantly improve infection control without being burdensome or overly expensive.
The fact that the testing progressed from cleaning in controlled environments to utilization of the Morantz Ultrasonics equipment in two different hospitals in England verified that the results represent a real-world application rather than just theoretical data. It should also be noted that the field testing in the hospitals was sponsored by the National Health Service in the United Kingdom as a demonstration project that was part of the Technology Innovation Program, which focuses specifically on reducing health care facility acquired infections.
A History of Effective Cleaning
Ultrasonic cleaners use sound waves produced at frequencies higher than our ears can perceive. The process involves the use of a generator, called a transducer, in a water tank, which creates high-frequency sound waves. As the sound waves move through the liquid they create compression waves that “tear” the liquid apart, leaving behind many millions of microscopic voids or partial vacuum bubbles. (The technical name for this effect is cavitation.) These small bubbles expand and eventually implode when they strike an object, and this energy dislodges contaminants even from intricately shaped surfaces.
Ultrasonic cleaning came into vogue for various types of industrial cleaning processes in the early 1950s. Small, table-top units quickly became common in jewelry and watch repair facilities, as well as in dental laboratories for cleaning surgical equipment. Many hospitals utilize ultrasonic cleaners for de-contaminating surgical instruments. The attraction of the ultrasonic cleaning process is that it minimizes the need for chemicals while allowing cleaning to occur in the smallest and hardest to reach places.
Large portable ultrasonic cleaning equipment was utilized in the 1970s for the cleaning of Venetian blinds, developed by Stan Morantz, one of the founders of Morantz Ultrasonics. From there, Morantz introduced the process to the developing restoration industry where it was found to be very effective at gently removing soot and fire residue from even the most fragile of items. Ultrasonic cleaning and Morantz took another leap forward at the end of the 20th century when it was shown to be effective at cleaning intricate electronic components, such as computer keyboards, without damage.
The usefulness of ultrasonic cleaning in regards to biological pathogens like bacteria and virus has been known for some time. However, until recently most of these efforts have been limited to small equipment such as clamps and dental molds. The testing done on the Morantz units for efficacy on large items such as wheelchairs, commodes, I.V. stands, bedside tables and other common hospital items is truly groundbreaking in the ultrasonic industry.
Not All Ultrasonic Equipment Is the Same
While all ultrasonic cleaners work on the same principle of cavitation, there are critical differences in the design and function of the various machines. Manufacturers generate sonic waves inside the tank of an ultrasonic cleaner in a variety of ways and in varying powers and frequencies. The testing showing the effectiveness of ultrasonic cleaning for controlling the spread of bacteria was done with equipment with specifications that included immersible transducer packs and a 40kHz frequency. Other manufactured machines have not been tested or proven to produce the same results.
Highlights of the Three Tests
In all three tests a common sense approach was followed. The ability of the Morantz ultrasonic equipment to kill microbial contaminants was measured by collecting samples from items prior to and after being cleaned. Immediate feedback was provided in all three studies by collecting samples on swabs that were analyzed by field equipment using adenosine triphosphate (ATP) technology. ATP instruments have a long history of use in food service and health care settings to determine the cleanliness of surfaces related to biological contaminants. Such instruments provide numerical results known as relative light units (RLU). In two of the studies, side-by-side surface samples were also analyzed by an independent laboratory to determine concentrations of specific types of bacteria.

The first independent test was conducted in order to determine if the cavitation process was as effective at removing bacteria as it was at dislodging dirt, grease, and other non-hazardous materials. A variety of residential and commercial items were tested, including toys, a wheelchair, and electronic components. The sampling data generated during the initial study revealed that:
- The Morantz ultrasonic cleaning system was extremely effective in removing bacteria from items with heavy contamination. Bacterial reduction of nearly 100% (99.86% and 99.98%, respectively) was achieved for items that started with more than 1 million and 259,900 colonies of bacteria, respectively.
- There was no evidence of cross-contamination even after the water in the ultrasonic tank had been used to clean items with extreme bacterial counts. In fact, one of the items cleaned immediately following contamination of the tank water by items with highly elevated bacterial concentrations had test results below the laboratory’s detection limit.

A follow-up study was authorized in an attempt to determine whether difficult-to-clean items from a hazardous environment could also be effectively decontaminated using a Morantz ultrasonic cleaner.
Since the testing of the Morantz ultrasonic equipment involved cleaning items that would be inoculated with dangerous bacteria (raw sewage and methicillin-resistant Staphylococcus aureus (MRSA)), appropriate precautions had to be taken. A small (~10′×10′×8′) enclosure was constructed with stud walls and double layers of six-mil plastic. A one-stage decontamination unit was attached to this enclosure to minimize the possibility of hazardous contaminants escaping from the test area. A HEPA-filtered negative air machine was utilized to keep the test area under negative pressure throughout the process.

On each item that was to be tested and cleaned, five squares were marked and numbered with indelible marker. All five areas on each item were contaminated in a uniform manner with either MRSA bacteria or raw sewage. Field samples were collected from each of the five test squares: three for analysis using a portable ATP tester (square 1, pre-cleaning; square 3, post-cleaning; square 5, post-cleaning and drying) and two for comparative laboratory analysis (square 2, pre-cleaning; square 4, post-cleaning and drying).
All items to be cleaned in the ultrasonic tank were intentionally contaminated prior to cleaning in order to ensure that the contents represented a worst-case scenario. Contamination of test items was conducted with commercially cultured methicillin-resistant Staphylococcus aureus bacteria or sewage obtained (post-screening) from the local wastewater treatment plant. As a further challenge, both porous and non-porous items were intentionally contaminated and then cleaned in the ultrasonic tank. Standard cleaning times (two to four minutes, depending on the object’s size, type, and material) were used so that results useful in real-world applications could be obtained.
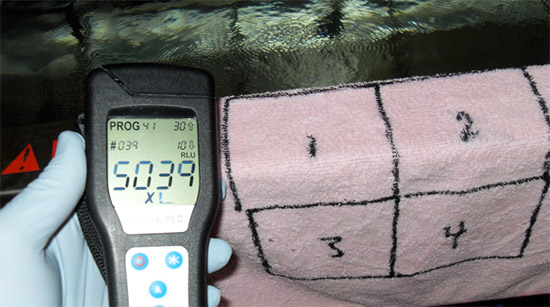
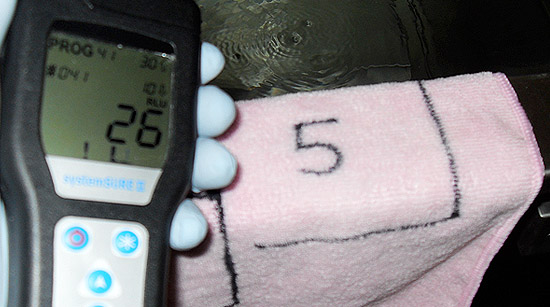
The sampling data generated during the second study showed that:
- Pernicious bacteria types, such as those resistant to antibiotics, can be effectively cleaned from surfaces using Morantz ultrasonic equipment, since MRSA bacteria were completely eliminated from contaminated surfaces by the ultrasonic cleaning.
- The laboratory results for Enterococcus and Escherichia coli bacteria contamination showed a 100% reduction for every non-porous item tested. The system also showed impressive results cleaning a microfiber cloth, with 100% reduction of E. coli bacteria and 97.7% reduction of colony forming units of Enterococcus.
- Laboratory testing revealed that live bacteria counts in the water when the ultrasonic machine was operating stayed close to zero even after intentional contamination with gallons of raw sewage.
MRSA bacteria was applied to the plastic tray of a walker using a swab.

Initial test results showed that the tray of the walker had been successfully contaminated as indicated by a reading of 925 relative light units (RLU).

As a comparison, the manufacturer of the ATP tester indicates that a clean surface would have less than 10 RLU’s.

Following a two-minute cleaning in the Morantz ultrasonic unit a sample was collected from square 3 and subjected to the same field analysis as the initial sample. Relative light units had been reduced from 925 to 10, indicating that the cleaning process was successful.

The two earlier studies garnered interest from the National Health Service in England as they aggressively attack the problem of HCAI. In this case a three-month trial was extended to six months because of the impressive results that were obtained. However, it is important to note that in the hospital study the “ultrasonics technology cleaning system was not intended to, and did not, replace standard cleaning. All equipment continued to be cleaned in the usual way”.(4) In other words, the infection control experts understand that ultrasonic cleaning is a supplement to existing infection control efforts, not a process that supplants it.
The British study was prompted by their understanding of the ultrasonic cleaning process:
Ultrasonic technology is good at accessing inaccessible areas that normal cleaning cannot reach, for example screw threads and hinges. It is good at cleaning hard substrates. It has been found by researchers to be even more effective than thorough hand scrubbing, often observed in busy work areas. It involves less exposure to cleaning agents and, therefore, contributes to a reduction in skin damage.(5)
1,025 measurements were collected from items before and after cleaning during the course of the study at the two hospitals. A careful analysis of the data showed “an average of a 98% change in the RLU reading when the average Pre-Clean reading is compared with the average Post-Clean 2 reading”.(6)

The results were so consistent and impressive that the researchers were able to draw a stronger correlation than expected. The stated goal was to measure the cleanliness of particular items with the expectation that better cleaning would, indeed, have a positive effect on the number of health care acquired infections. At the conclusion of the trial period the study authors noted, “The evaluation was not designed to assess the effectiveness of ultrasonics in reducing infection, but was it more effective than normal routine cleaning”.
As part of the study in England, hospital personnel were asked to evaluate the ultrasonic cleaning process. When those individuals who had knowledge of the trial were asked, “Would you recommend the system to other colleagues?” 100% of the responses were positive.(7)
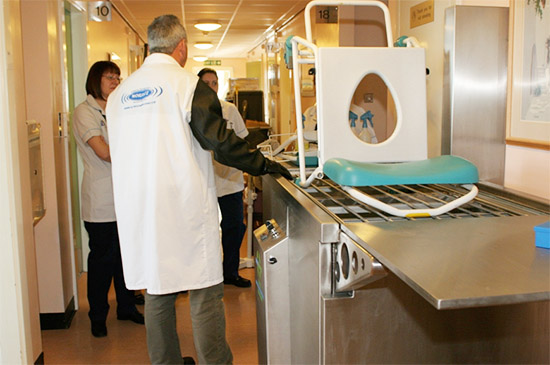
At the conclusion of the study hospital personnel had some specific recommendations and conclusions.(8) They noted that the ultrasonic cleaning was especially useful for:

Overall, the study authors were enthusiastic about the Morantz ultrasonic cleaning equipment as a new weapon in the war on HCAI as they stated:
The…ultrasonic system can be used without disruption to staff or patients so long as there is a plan in place to ensure equipment that may be required frequently through the day is cleaned at an appropriate time to allow it to be returned before being required again. The clean appearance of the medical equipment returned had a lot of support from staff and the RLU values post pre clean proves equipment was less contaminated.(9)
An Ultrasonic Answer to Controlling Infections
Both controlled testing and field demonstrations have confirmed that Morantz ultrasonic cleaning equipment is an important tool that should be utilized to improve cleaning performance in health care settings and other critical use facilities. This improved cleaning is one of the keys to reducing health care acquired infections and limiting the personal suffering and financial loss that comes from these preventable diseases.
- Monina Klevens, DDS, MPH et. al., “Estimating Health Care-Associated Infections and Deaths in U.S. Hospitals 2002,” Public Health Reports, Centers for Disease Control and Prevention, March–April 2007, Volume 122.
- Douglas Scott II, “The Direct Medical Costs of Healthcare-Associated Infections in U.S. Hospitals and the Benefits of Prevention,” Division of Healthcare Quality Promotion National Center for Preparedness, Detection, and Control of Infectious Diseases; Coordinating Center for Infectious Diseases; Centers for Disease Control and Prevention, March 2009.
- Baubiology is the study of how buildings impact the people who occupy them. It is not limited only to the practice of reducing toxicity of building materials, but represents a total approach to a healthy living environment. The approach of baubiology examines lessons from the past, but is future focused. It emphasizes that a particular combination of building materials, furnishings, and cleaning can create positive or negative synergies.
- “Showcase Hospitals Local Technology Review Report number 5: Bio-Cav40 Ultrasonic Cleaning,” Calderdale and Huddersfield NHS Foundation Trust, p. 10.
- Ibid., p. 8.
- Ibid., p. 12.
- Ibid., p. 16.
- Ibid., p. 7.
- Ibid., p. 18.
ABOUT THE AUTHOR

Michael A. Pinto, CSP, CMP, is chief executive officer of Wonder Makers Environmental, Inc. He focuses on common sense practices that work when health professionals are faced with threats from swine or avian flu, MRSA outbreaks, norovirus exposures, and nosocomial infection problems in healthcare facilities.
Mr. Pinto is the author of over 150 published articles and several books including, Fungal Contamination: A Comprehensive Guide for Remediation. He completed doctoral course work in environmental engineering and holds numerous certifications in the environmental and safety areas including Certified Safety Professional and Certified Mold Professional. He conducted most of the testing described in this article but has no ownership connection to Morantz or other manufacturers of mentioned products.
